|
TEST DRIVE: Abbott's FreeStyle Libre - Transforming Glucose Monitoring Through Utter Simplicity, Fingersticks Aside! – An in-depth overview of the FreeStyle Libre system, plus how it compares to CGMs.įreeStyle Libre Pro Submitted for FDA approval, Potentially Coming to the US in 2016 – New blinded sensor worn for 14 days that will give a detailed picture of your glucose levels.Ĭomparing FreeStyle Libre vs. Sensor patch – worn on the upper arm for 14 daysĪbbott Debuts Flash Glucose Monitoring System: A New Alternative to Fingersticks– An article on a new alternative to fingersticks and CGM’s.įreeStyle Libre Approved in Europe – An detailed overview of the Freestyle Libre system, including design, pricing, and accuracy data. Touchscreen reader device – displays data and trend information To use the FreeStyle Libre, multiple parts are required: The FreeStyle Libre system does not require fingerstick calibration, so users can dose insulin based on its readings (except for when hypoglycemic, when glucose levels are rapidly changing, or when symptoms don’t match the system’s readings). However, unlike CGM, FGM does not have hypo- or hyperglycemia alarms and will only provide a trend graph if it has been swiped in the past eight hours. This allows people to get individual blood sugar readings (like BGM) and trend information (like CGM). When the reader device is swiped close to the sensor, the sensor transmits both an instantaneous glucose level and eight-hour trend graph to the reader. In Flash Glucose Monitoring, patients have a sensor inserted on their upper arm and a separate touchscreen reader device. The Abbott FreeStyle Libre is currently the only flash glucose monitoring product available, and it is currently only approved in Europe. FreeStyle, Libre and related brand marks are marks of Abbott.What It Does: Flash Glucose Monitoring* (FGM) is the newest method of glucose testing that is seen as a hybrid between meters and CGMs. All trademarks referenced are trademarks of either the Abbott group of companies or their respective owners. For additional information, please read the user’s manual. 
The Sensor must be removed prior to Magnetic Resonance Imaging (MRI). This material is intended for Healthcare Professionals. 
Product is in Compliance with the applicable regulation on medical devices. Not all features are available in all markets. Product availability may vary by country. If you live in another country, please contact your Local Abbott office to obtain the correct product information for your country of residence.įreeStyle Libre Portfolio is available in both mmol/L and mg/dl versions depending on the country. This is a product specific website intended for Health Care Providers of the European Union only. Sensor is water resistant in up to 1 metre (3 feet) of water for a maximum of 30 minutes. 60-minute warm-up required when applying the sensor.Ĩ. Dexcom G6 CGM User Guide and Medtronic Guardian Connect System User Guide.ħ. “Accuracy of a 14-Day Factory-Calibrated Continuous Glucose Monitoring System With Advanced Algorithm in Pediatric and Adult Population With Diabetes.” Journal of Diabetes Science and Technology, (September 2020).Ħ. Christiansen, Gregory Forlenza, Mark Kipnes, David R. 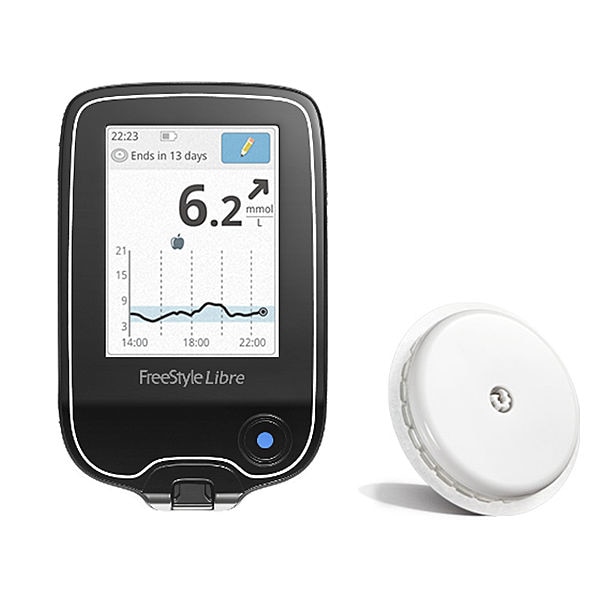
Alva, Shridhara, Timothy Bailey, Ronald Brazg, Erwin S. Data on file, Abbott Diabetes Care, Inc.ĥ. Use of FreeStyle LibreLink requires registration with LibreView.Ĥ. 
Please check the website for more information about device compatibility before using the app. The FreeStyle LibreLink app is only compatible with certain mobile devices and operating systems. Once the patient scans their FreeStyle Libre 2 sensor with that device, they can receive alarms only on that device. They must start their FreeStyle Libre 2 sensor with that selected device. Patients choose which device they want to receive alarms: FreeStyle Libre 2 reader or FreeStyle LibreLink app. Finger pricks are required if your glucose readings and alarms do not match symptoms or expectations.ģ. Data based on the number of users worldwide for the FreeStyle Libre system compared to the number of users for other leading personal use sensor based glucose monitoring systems.Ģ. Images are for illustrative purposes only.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
